Efficacy
The efficacy of BRUKINSA®▼ for the treatment of WM was evaluated in a randomised trial:1,2
- ASPEN was a phase 3, open-label, randomised multicentre trial comparing BRUKINSA with ibrutinib in patients with WM who were Bruton’s tyrosine kinase (BTK) inhibitor naïve1–4
Results from the ASPEN study demonstrate that BRUKINSA is an effective treatment for WM.2 Treatment with BRUKINSA was associated with a trend towards better disease control and greater improvement in quality of life measures versus ibrutinib.2
Very good partial response (VGPR) or complete response (CR)
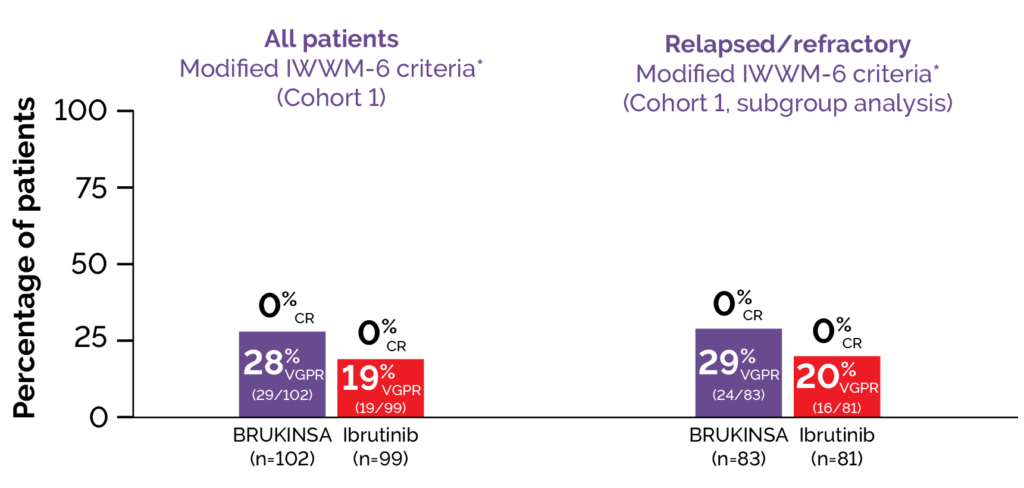
In the ASPEN study, the primary end point was the proportion of patients in Cohort 1 (MYD88L265P) who achieved a VGPR or CR, as assessed by an independent review committee (IRC).2 The testing for the superiority of the primary endpoint required testing in the relapsed/refractory (R/R) analysis set prior to testing in the intention-to-treat (ITT) analysis set.1,2
At a median follow up of 19.4 months, 29% (24/83) of patients with R/R disease in the BRUKINSA arm, and 20% (16/81) in the ibrutinib treatment arm achieved VGPR (2-sided p=0.12; Figure 1), therefore the primary endpoint was not significant in the R/R analysis set.2 In the ITT analysis set, a higher proportion of patients in the BRUKINSA treatment arm achieved VGPR compared with the ibrutinib arm (28% [29/102] versus 19% [19/99], respectively; 2-sided p=0.09; Figure 1).2 This trend was also observed amongst treatment-naïve patients (26% [5/19] versus 17% [3/18] in the BRUKINSA and ibrutinib treatment arms respectively; 2-sided p=0.54). No patients in either arm of Cohort 1 achieved a CR (N=201).2 Extended follow-up results at median 44.4 months showed that VGPR rates increased over time and were numerically higher with BRUKINSA than ibrutinib at all time points.4 VGPR rates were 36.3% with BRUKINSA and 25.3% with ibrutinib.4
Patients assigned to Cohort 2 (MYD88WT) received BRUKINSA; after a median follow up of 17.9 months, 27% (7/26) of patients had achieved a VGPR.3 After a median follow up of 30 months the VGPR + CR rate was 30.8% with one CR.4
Figure 1. ASPEN: Patients with IRC-assessed VGPR in BRUKINSA versus ibrutinib treatment arms.2
Major response rate (MRR)
MRRs were similar between treatment groups.2 MRRs overall were 77% (95% CI 68, 85) and 78% (95% CI 68, 86) in the BRUKINSA and ibrutinib treatment arms respectively.2 MRRs for treatment-naïve patients were 74% (95% CI 49, 91) and 67% (95% CI 41, 87), and for patients with R/R disease were 78% (95% CI 68, 87) and 80% (95% CI 70, 88) in the BRUKINSA and ibrutinib treatment arms respectively.2 MRR was 50% (95% CI 30, 70) for patients in Cohort 2.1,3
After extended follow-up (median 44.4 months for Cohort 1 and 42.9 months for Cohort 2), MRRs were 79.8% (95% CI 70.5, 87.2) and 81.4% (95% CI 72.4, 88.4) with BRUKINSA and ibrutinib respectively in Cohort 1, and 65.4% (95% CI 44.3, 82.8) for Cohort 2.4
Progression-free survival (PFS)
After a median follow-up of 18 and 18.5 months, 15% of patients treated with BRUKINSA and 16% of patients treated with ibrutinib in Cohort 1 progressed or died, and median PFS was not reached for either arm.2 After a median follow-up of 17.9 months, median PFS was not reached in Cohort 2.3
Based on an updated data cut-off at 30 months, the progression free-survival event-free rate by investigator assessment was 84.9% versus 77.6% for BRUKINSA versus ibrutinib respectively, with an estimated overall hazard ratio of 0.734 (95% CI: 0.380, 1.415).1 After extended follow-up (median 44.4 months for Cohort 1 and 42.9 months for Cohort 2) median PFS was not reached for either treatment arm in Cohort 1 and median PFS was 45.8 in Cohort 2.4
Abbreviations: CI, confidence interval.
